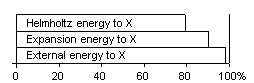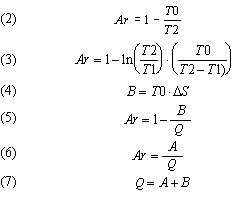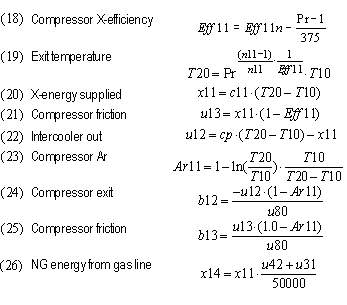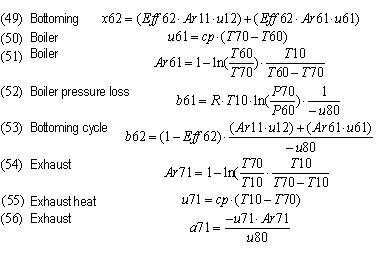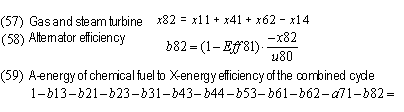Second law Analysis of Gas Turbines
|
CONTENTS 12pg 45K 11fig 1. Abstract 2. Background 3. Focus 4. The plural energy system 5. Energy vs work and heat 6. Thermal temperature and chemical temperature 7. Splitting up the exergy concept 8. The carnot ratio and helmholtz ratio 9. Polytropic gas turbine analysis 10. Six open cycle gas turbines analyzed 11. Inputs for metal 49MW 4Pr gas turbine 12. Compressor 13. Heat exchanger 14. Turbine 15. Combustion 16. Bottoming cycle and exhaust 17. Efficiency of total cycle 18. Reduction of combustion irreversibility's 19. Conclusions 20. References 21. Symbols 22. Notes 23. Revision history >>>Copyright 2023Mar05 by Ben Wiens...energy scientist
1. ABSTRACT
2. BACKGROUND
3. FOCUS
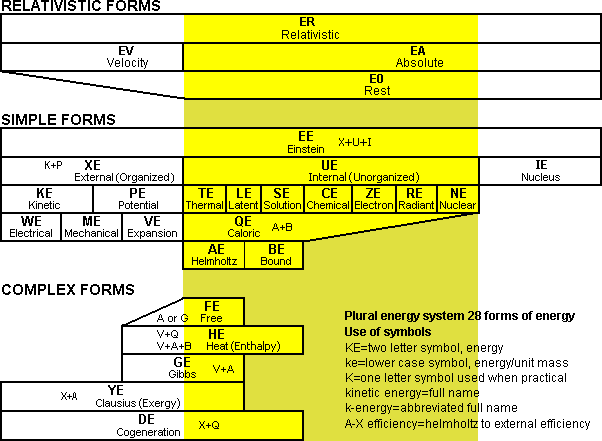
4. THE PLURAL ENERGY SYSTEM (C1)
Fig 1 Bar chart illustrating the plural energy system
The first concept that will be introduced is the plural energy system as shown in Fig 1. Such a system was started in the 1800s by Gibbs and especially Helmholtz who it appears also viewed these terms as real subforms of energy (Bailyn 1994). In this system, the concept of exergy is replaced by clausius energy ...is a specific form of energy composed of external energy and helmholtz energy. There are 28 forms of energy shown in total. A key advantage of the plural energy system is that it allows the present exergy evaluation to be broken into eleven divisions; external, kinetic, potential, electrical, mechanical, expansion, helmholtz, free, gibbs, clausius, and bound energy. This makes a second law evaluation more precise as each of these are not really equal in worth when comparing realistic processes.
5. ENERGY vs WORK AND HEAT (C2)
6. THERMAL TEMPERATURE AND CHEMICAL TEMPERATURE (C3) 7. SPLITTING UP THE EXERGY CONCEPT (C4)
In the singular energy system, all more specific forms of clausius energy are generally lumped together. The typical exergy equation (1) does start with five different types of energy in the equation but then adds them together.
Fig 2 Typical efficiency of conversion to X-energy in GT 8. THE CARNOT RATIO AND HELMHOLTZ RATIO (C5, 6, 7)
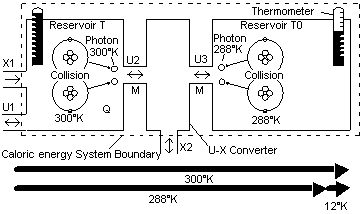
Fig 3 System diagram for conversion of energy
Entropy is used extensively in a typical exergy evaluation to calculate the bound energy produced. In Eq (1) this is the TS term. As can be seen, the exergy equation is an additive-subtractive equation.
Fig 4 Carnot ratio diagram for Metal 4Pr of Fig 6
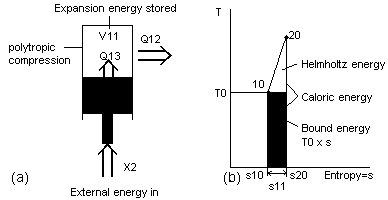
Because many processes occur over varying temperatures, it is helmholtz energy A and the helmholtz ratio Ar that is mostly used. It is important to think of helmholtz energy A and bound energy B in the right way. The helmholtz energy A is the part of caloric energy Q that could be converted into external energy X in a future process. The bound energy B is the caloric energy Q that could have been converted into external energy X if the conversion had started at an infinite temperature and progressed to the present point.
10. SIX OPEN CYCLE GAS TURBINES ANALYZED (C9)
11. INPUTS FOR METAL 49MW 4PR GAS TURBINE
Fig 5 Differences between ratio and subtractive calculations
To model the gas turbine compression process, a piston in a cylinder is used as shown in Fig 5a. The compression is an inefficient polytropic process which would ideally model the intercooled compressor of the WR-21 powerplant. In the proposed method of analyzing such a process, the external energy X2 that is supplied to the system is converted into three new separate streams of energy. There is expansion energy V11 which is stored inside the cylinder, caloric energy Q13 due to friction which is also stored in the cylinder and caloric energy Q12 that is created due to transfer out of the cylinder.
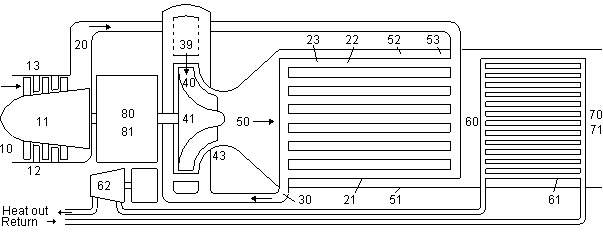
Fig 6 Gas turbine model used in Table 1 |
4Pr 4Pr Inputs ex=exchanged comb=combined cycle | Symbol | ex, comb ex, comb comb comb ex simple Nominal compressor efficiency at Pr=2 | Eff11n | 0.92 | 0.92 | 0.90 | 0.92 | 0.88 | 0.86
| Nominal turbine efficiency at Pr=2 | Eff41n | 0.92 | 0.92 | 0.90 | 0.92 | 0.88 | 0.96
| Compressor exponent | n11 | 1.39 | 1.39 | 1.39 | 1.39 | 1.24 | 1.39
| Turbine exponent | n41 | 1.25 | 1.30 | 1.33 | 1.37 | 1.33 | 1.30
| Compressor inlet temperature | T10 | 288°K | 288°K | 288°K | 288°K | 288°K | 288°K
| Combustion exit temperature | T39 | 1973°K | 1223°K | 1623°K | 1623°K | 1437°K | 1100°K
| Boiler exit temperature | T70 | 348°K | 348°K | 348°K | 422°K | 348°K | 348°K
| Compressor pressure ratio | Pr | 4.0 | 4.0 | 16.6 | 29.5 | 14.0 | 6.5
| Exchanger high pressure loss | L21 | 0.035 | 0.035 | - | - | 0.10 | -
| Combustor pressure loss | L31 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05
| Exchanger low pressure loss | L51 | 0.02 | 0.02 | 0 | 0 | 0.05 | 0
| Boiler low pressure loss | L61 | 0.02 | 0.02 | 0.02 | 0.02 | 0 | 0
| Exchanger effectiveness at equal flow | Eff21 | 0.975 | 0.95 | - | - | 0.88 | -
| Bottoming conversion of exhaust A-energy | Eff62 | 0.71 | 0.71 | 0.71 | 0.60 | - | -
| Air cooling bypass % of compressor | By41 | 0 | 0.00 | 0.15 | 0.10 | 0.15 | 0
| Alternator efficiency | Eff81 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.95
| Calculations | - | - | - | - | - | - | -
| Adjusted compressor Eff due to Pr and size | Eff11 | 0.912 | 0.912 | 0.878 | 0.844 | 0.845 | 0.845
| Compressor exit temperature | T20 | 441°K | 441°K | 706°K | 887°K | 527°K | 536°K
| Equivalent turbine inlet temperature no cooling | T40 | 1973°K | 1223°K | 1486°K | 1549°K | 1301°K | 1100°K
| Adjusted turbine efficiency due to Pr and size | Eff41 | 0.904 | 0.912 | 0.853 | 0.827 | 0.820 | 0.830
| Turbine exit temperature | T50 | 1549°K | 925°K | 809°K | 720°K | 757°K | 765°K
| Adjusted heat exchanger effectiveness | Eff22 | 0.975 | 0.950 | - | - | 0.748 | -
| Total internal energy added | u80 | 637.9 | 395.8 | 893.0 | 722.2 | 670.0 | 650.0
| External energy from bottoming | x62 | 25.4 | 24.4 | 157.7 | 89.9 | - | -
| Total external energy extracted | x82 | 478.8 | 239.3 | 519.4 | 367.2 | 278.8 | 166.6
| Helmholtz ratio | - | - | - | - | - | - | -
| Compressor | Ar11 | 19.8% | 19.8% | 38.2% | 45.9% | 27.2% | 27.9%
| Combustion of fuel | Ar31 | 83.4% | 72.7% | 72.5% | 75.7% | 70.8% | 63.3%
| Turbine | Ar41 | 83.6% | 73.0% | 74.1% | 73.4% | 71.3% | 68.8%
| Exchanger out | Ar22 | 68.1% | 57.0% | - | - | 55.7% | -
| Exchanger in | Ar52 | 67.0% | 55.3% | - | - | 53.7% | -
| Boiler | Ar61 | 29.0% | 28.7% | 47.3% | 48.4% | 35.0% | 45.6%
| Exhaust | Ar71 | 9.2% | 9.2% | 9.2% | 17.9% | 9.2% | 9.20%
| A-B-energy loss % of fuel A-energy | - | - | - | - | - | - | -
| Compressor friction | b13 | 1.73% | 2.79% | 3.60% | 7.16% | 6.04% | 4.35%
| Exchanger slippage | b21 | 1.94% | 1.97% | 0 | 0 | 0.61% | 0
| Exchanger high pressure | b23 | 0.11% | 0.18% | 0 | 0 | 0.09% | 0
| Combustion | b31 | 16.48% | 27.18% | 27.06% | 24.10% | 28.90% | 35.85%
| Turbine friction | b43 | 1.51% | 2.44% | 3.14% | 6.00% | 4.99% | 3.67%
| Combustion pressure drop | b44 | 0.16% | 0.26% | 0.03% | 0.02% | 0.04% | 0.09%
| Exchanger low pressure | b53 | 0.25% | 0.40% | 0 | 0 | 0.61% | 0
| Bottoming cycle or exhaust loss no bottoming | b62 | 1.62% | 2.52% | 7.21% | 8.30% | 15.70% | 29.55%
| Boiler pressure drop | b61 | 0.26% | 0.41% | 0.12% | 0.23% | 0 | 0
| Exhaust | a71 | 0.87% | 1.40% | 0.62% | 3.33% | 0.84% | 0.85%
| Generator | b82 | 0.75% | 0.60% | 0.58% | 0.51% | 0.42% | 1.28%
| Powerplant fuel A-X efficiency | - | 74.30% | 59.84% | 57.58% | 50.34% | 41.76% | 24.35%
| Cogeneration fuel A-Y efficiency | - | 75.17% | 61.23% | 58.20% | 53.67% | 58.14% | 53.27%
| |
|
Table 1 Comparison of six gas turbines
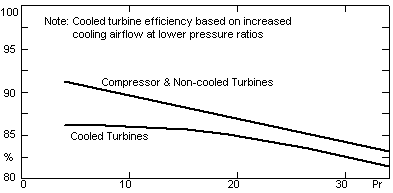
One of the problems with higher pressure ratios is that the compressor and turbine polytropic efficiency becomes less (C10). Equation (18, 37) adjust for this and are plotted in Fig 7. Single spool engines like the Siemens V84.3A are most affected and multi-spool like the GE S-160 are less affected (Wilson 1984).
Fig 7 X-V efficiencies with increasing pressure ratios
Not only can the heat exchanger reduce bound energy B production in the combustion chamber, it is also able to recycle much of the thermal energy T in the exhaust to the incoming air. There is a limit to how much of the exhaust can be recycled because of the compressor exit temperature. The compressor can be intercooled, but calculations showed that using a bottoming cycle steam turbine instead of one intercooling stage on the Metal 4Pr resulted in higher efficiencies in large powerplants.
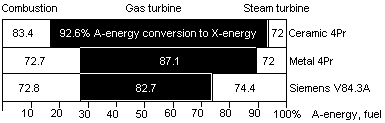
Fig 8 A-X efficiency in 3 gas turbines of Table 1
In a heat exchanged gas turbine the hot gases are only expanded slightly before the remaining thermal energy T is recycled to the incoming gases. This would seem wasteful, however maintaining a high temperature in the turbine results in a high ratio of turbine to compressor work and results in a higher A-X efficiency of the combined compressor and turbine unit.
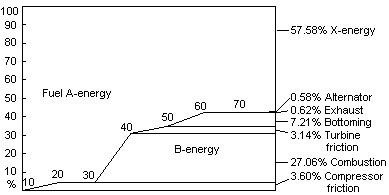
Fig 9 Fuel A-energy to X-energy in Siemens V84.3A
It is well known that the single largest production of bound energy B in the gas turbine occurs due to necessary cooling of gases in the combustion chamber and first stages of the turbine. For an advanced combined cycle such as the Siemens V84.3A, the compressor, turbine, bottoming cycle and exhaust bound energy B created is small compared to this cooling bound energy B created. This is illustrated in Fig 9. Production of bound energy B in the combustion chamber occurs because the chemical energy C has been converted to thermal energy T at a lower temperature than the maximum possible for the particular fuel used. Methane has a helmholtz ratio of 91.9% based on the higher heating value (hhv). If this is plotted on the carnot ratio diagram of Fig 4, this equals a mean temperature in Reservoir T of 3556°K, based on a 288°K ambient temperature. In the Siemens V84.3A the mean temperature of combustion however is only 1048°K which equals a helmholtz ratio Ar of 72.5%. The bound energy B produced by this difference in helmholtz ratios Ar is calculated with Eq (48). Note that in Table 1 the cooling losses of the turbine are approximated by creating an adjusted maximum combustion temperature in Eq (36).
Fig 10 Energy transformer model
It is known that electrochemical fuel cells can avoid combustion irreversibility's by converting chemical energy C directly into electricity. Real fuel cells though have considerable internal resistance and so produce thermal energy T as a by-product. Presently the best large fuel cell powerplants must use bottoming cycle engines to reach their A-X net electrical efficiency of 57% (Kordesch 1996) when operating at rated power. If the combustion irreversibility's are not considered, the Siemens V84.3A gas turbine combined cycle powerplant has an A-X net electrical efficiency of 85%. This indicates that the gas turbine type powerplant after the combustion process is inherently more efficient at converting helmholtz energy A than the fuel cell. Indeed Fig 8 shows that the A-X conversion efficiency of the compressor-expander portion of the gas turbine could approach 93% A-X efficiency in future high temperature gas turbine powerplants. Perhaps the fuel cell should be rather optimized as a gas turbine topping cycle. In the future it may be possible to design a very high temperature Solid Oxide fuel cell which would supply thermal energy to the Ceramic 4Pr gas turbine at up to 1973°K. A real powerplant A-X efficiency of up to 88% may be possible.
19. CONCLUSIONS
20. REFERENCES
21. SYMBOLS
22. NOTES
23. REVISION HISTORY
COPYRIGHT © 1999-2023 All rights reserved |